 This video shows you how to draw the different molecular orbitals for a conjugated system, how to make sense of the logic without the crazy math, and how to quickly and easily identify HOMO and LUMO molecular orbitals. 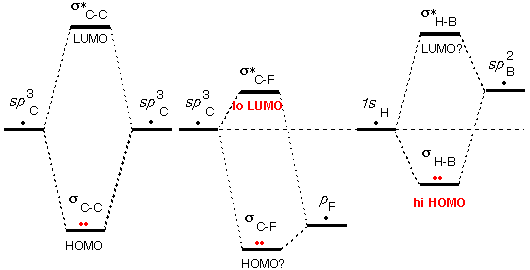
Video 2: HOMO and LUMO for Conjugated Systems Few approaches can simultaneously lower LUMO and HOMO energy levels of conjugated polymers to a large extent (>0.5 eV). This video provides you with a logic-based overview of MO theory with simple take-aways to focus on at the organic chemistry level. The problem in the log file is that the 10 lowest eigenvalues of the unoccupied subspace are quite larger than the 10. The key parameters of conjugated polymers are lowest unoccupied molecular orbital (LUMO) and highest occupied molecular orbital (HOMO) energy levels. In the HOMO level, the distribution of the positive and negative linear combination of atomic orbital (LCAO) coefficients shows a change in the sign of the wave function every half monomer unit (see Fig. Video 1: Molecular Orbital Theory for Sigma and Pi Bonds The calculated evolutions can be rationalized from the shape of the HOMO and LUMO orbitals of a single 6T molecule. 
MO theory typically comes up when studying reactions of conjugated systems, especially when asked to identify HOMO and LUMO molecular orbitals for molecules in Diels-Alder and other pericyclic reactions. 
While thinking of the valence and conduction band edges as the 'HOMO or LUMO of a crystal' is often quite fruitful, there are some effects present in solids that cannot be understood by this simple pov. Molecular Orbital theory is a topic that comes up in general chemistry in relation to bond formation and bonding energy.Īt the organic chemistry level, we’re less concerned with the nitty-gritty quantum physics and crazy math and more interested in making just enough sense of the information to be able to apply what we know to reactions and mechanisms. maximal positive overlap between the highest occupied molecular orbital HOMO and the lowest unoccupied molecular orbital LUMO. A HOMO/LUMO is just a single energy level. Comparison Study of HOMO-LUMO Energy Gaps for Tautomerism of Triazoles in Different Solvents Using Theoretical Calculations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
